
Benign Is Not Fine. It’s Time to Rally for Research
May is Brain Tumor Awareness Month, and this month the Raymond A. Wood Foundation is ... Read more

Advancing Understanding of Hyperphagia in CP
New Study Sheds Light on Hyperphagia in Craniopharyngioma We’re thrilled to share the publication of ... Read more

Team RAWF Takes on NYC to Support Brain Tumor Survivors!
The streets of New York City were full of energy, optimism and determination as a dedicated group ... Read more
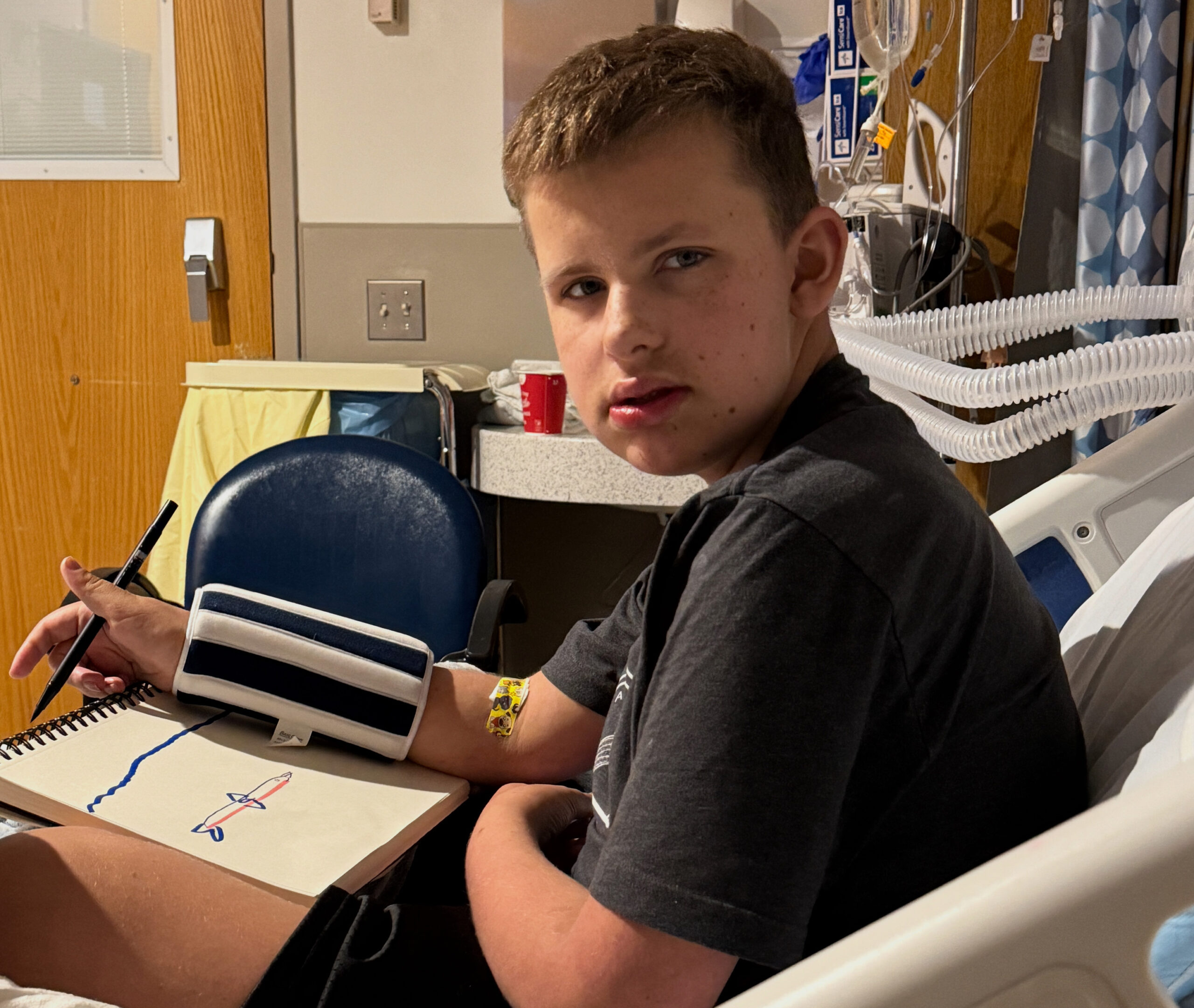
Hypothalamic Syndrome: Finally, a Way to Describe What We’ve Been Living
Recognizing hypothalamic syndrome offers new hope for understanding, preventing, and treating the complex challenges faced ... Read more

Medical ID Bracelets for Panhypopituitarism Patients
It’s a common question that comes up among Facebook support groups for brain tumor patients ... Read more

The Raymond A. Wood Foundation and NORD® Launch Natural History Study of Rare Hypothalamic-Pituitary Brain tumors.
Research study is open to participants worldwide to advance understanding and treatments these rare diseases ... Read more

Clinical Trial Participant Best Practices
As a Participant, it’s Important to Do Your Part to Maintain the Integrity of the ... Read more

Raymond A. Wood Foundation Reveals Caregiver Burden in Pioneering Craniopharyngioma Study
The Raymond A. Wood Foundation recently published its first research study entitled “Caregiver burden and ... Read more

Navigating Special Education for a Successful School Year
In preparation of the upcoming school year, we dive into a crucial topic for parents ... Read more

RAWF Executive Director at the IBTA World Summit in Vienna, Austria
Brain tumor patient advocates, clinicians and researchers convened in Vienna, Austria last month at the ... Read more
