by Nathalie Kayadjanian, Ph.D., RAWF Scientific Advisor
Image Credit: National Cancer Institute
While surgery and radiotherapy represent the recommended first-line therapies for CP, they are responsible, in part, for the co-morbidities associated with CP including primary metabolic alterations, such as obesity, neuro-endocrine disorders, and psycho-social alterations. Recent progress made in the understanding of the genomic alterations and molecular pathways involved in CP tumorigenesis have contributed to advancing diagnostic and testing drugs targeting these pathways. One successful example is the significant tumor reduction measured in patients with an adult-onset CP treated with BRAF and MEK inhibitors reported recently in several clinical studies and a phase II clinical trial (Fasano et al., 2022). The detection of BRAF mutations in serum (Brastianos et al.2016) may contribute to preoperative diagnostic evaluations in the near future. Altogether, these results are paving the way for non-operative diagnostic and neoadjuvant molecular-targeted treatment strategy for CP patients.
Growth hormone (GH) deficiency affects growth pattern in 75% of children with CP. In adults with CP, GH deficiency is characterized by decreased muscle mass, increased body fat, reduced energy, and well-being. GH replacement therapy is the only effective therapy. Besides its positive effect on growth in children, GH replacement has benefits on body composition, bone health, metabolic outcomes, and quality of life. Although GH therapy raised safety concerns due to its known pro-proliferative, angiogenic, and anti-apoptotic properties, numerous studies have demonstrated that GH replacement therapy does not increase the risk of tumor recurrence in patients with CP. This year, a consensus statement in favor of GH therapy in CP has been published (Boguszewski et al., 2022).
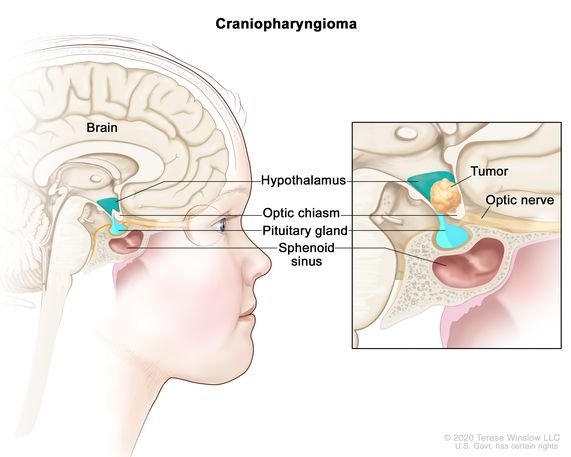
Two types of CP, the adamantinomatous (ACP) and papillary (PCP) have been previously described based on histological features. Thanks to molecular and clinical advances, WHO changed the taxonomy on CNS brain tumors (5th edition), so that, ACP and PCP are now recognized as distinct CP tumor types, given their different clinicodemographic, radiologic, histopathologic, and genetic alteration profiles. Although it can affect adults, ACP is mostly a pediatric tumor and represents the most common pituitary tumor in children. Childhood Craniopharyngioma treatment focuses on balancing effective tumor removal with the preservation of hormonal function and quality of life. In contrast, PCP affects predominantly adults.
CP research is lacking evidence-based data on the viewpoints of patients and caregivers on symptoms of importance and treatment priorities. A survey of patients and caregivers has been recently published to better understand the approaches that families take towards weight management and the impact on social function in individuals with CP and obesity (Craven et al., 2022). Of interest, the findings highlight the opportunity to improve social functioning as an additional potential benefit of improved treatments for hypothalamic obesity.
Paralleling the improvements in surgical outcomes, the standard for successful surgery has expanded to focus on patient-reported quality of life (QoL). A recent review has shown that the QoL of the patients with CP was lower than that of the general population (Castle-Kirszbaum et al., 2022). Hypothalamic involvement was consistently the strongest predictor of QoL. Endocrinopathy contributed to morbidity but could be ameliorated by hormone replacement therapy. Social and emotional dysregulation and a poor memory are common complaints after surgery. This study shed lights on understudied comorbidities that affect significantly the QOL of patients with CP.
Sleep disorders represent an important issue in patients with CP. They remain, however, largely underdiagnosed and untreated in clinical practice and no guidelines are available for the diagnosis and management of sleep disorders in these patients. Romigi and coll. (2022) have published a recent paper on the current knowledge of the pathogenesis and prevalence of sleep disorders in patients with CP and propose practical algorithms for their evaluation and management in clinical practice.
Related Links:
Current craniopharyngioma clinical trials
A Survey of Patient-Relevant Outcomes in Pediatric Craniopharyngioma: Focus on Hypothalamic Obesity
Resources for newly diagnosed
References
Boguszewski MCS et al., (2022). Safety of growth hormone replacement in survivors of cancer and intracranial and pituitary tumours: a consensus statement. Eur J Endocrinol. 2022 Apr 21;186(6):P35-P52.
Castle-Kirszbaum M, et al., (2022). Quality of Life in Craniopharyngioma: A Systematic Review. World Neurosurg. Aug;164:424-435.e2. doi: 10.1016/j.wneu.2022.05.038.
Craven M, et al (2022) A Survey of Patient-Relevant Outcomes in Pediatric Craniopharyngioma: Focus on Hypothalamic Obesity. Front. Endocrinol. 13:876770.
Fasano M et al., (2022) Dramatic Therapeutic Response to Dabrafenib Plus Trametinib in BRAFV600E Mutated Papillary Craniopharyngiomas: A Case Report and Literature Review. Front. Med. 8:652005.
Romigi A. et al., (2022), Sleep Disorders in Patients with Craniopharyngioma: A Physiopathological and Practical Update. Front Neurol. 2022 Feb 9;12:817257. doi: 10.3389/fneur.2021.817257.